|
11/1/2022 0 Comments Nucleus atom 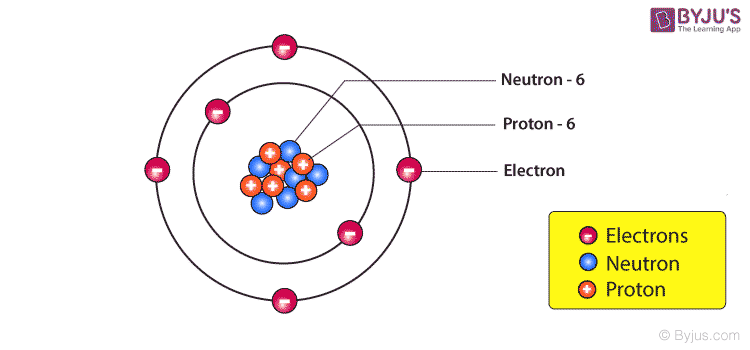 /GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)
The combined mass of the electrons is very small in comparison to the mass of the nucleus, since protons and neutrons weigh roughly 2000 times more than electrons. Protons and neutrons have nearly equal masses, and their combined number, the mass number, is approximately equal to the atomic mass of an atom. The number of protons and neutrons together determine the nuclide (type of nucleus). The first method is used in producing enriched uranium from a sample of regular uranium, and the second is used in carbon dating. Different isotopes in a sample of a particular chemical can be separated by using a centrifuge or by using a mass spectrometer. Different isotopes of the same element have very similar chemical properties. The isotope of an atom is determined by the number of neutrons in the nucleus. Hence, the atom is made up of mostly empty space. This is comparable to a fly in a cathedral. The size of a nucleus is of the order of 10 − 15m compared to the atom, which is of the order 10 − 10m. Thomson found the electron, through an expirement in which he used a tube with a cathode ray. Lewis stated, in his famous article The Atom and the Molecule, that “the atom is composed of the kernel and an outer atom or shell”. The adoption of the term “nucleus” to atomic theory, however, was not immediate. The modern atomic meaning was proposed by Ernest Rutherford in 1912. In 1844, Michael Faraday used the term to refer to the “central point of an atom”. The etymology of the term nucleus is from 1704 meaning “kernel of a nut”. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus with a very small contribution from the orbiting electrons. These dimensions are much smaller than the size of the atom itself by a factor of about 23,000 (uranium) to about 145,000 (hydrogen). The size (diameter) of the nucleus is in the range of 1.6 fm (10 -15 m) (for a proton in light hydrogen) to about 15 fm (for the heaviest atoms, such as uranium). The nucleus of an atom is the very small dense region of an atom, in its center consisting of nucleons ( protons and neutrons). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
